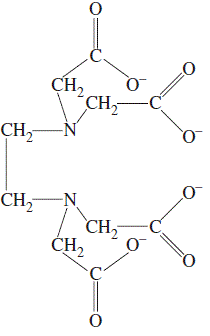
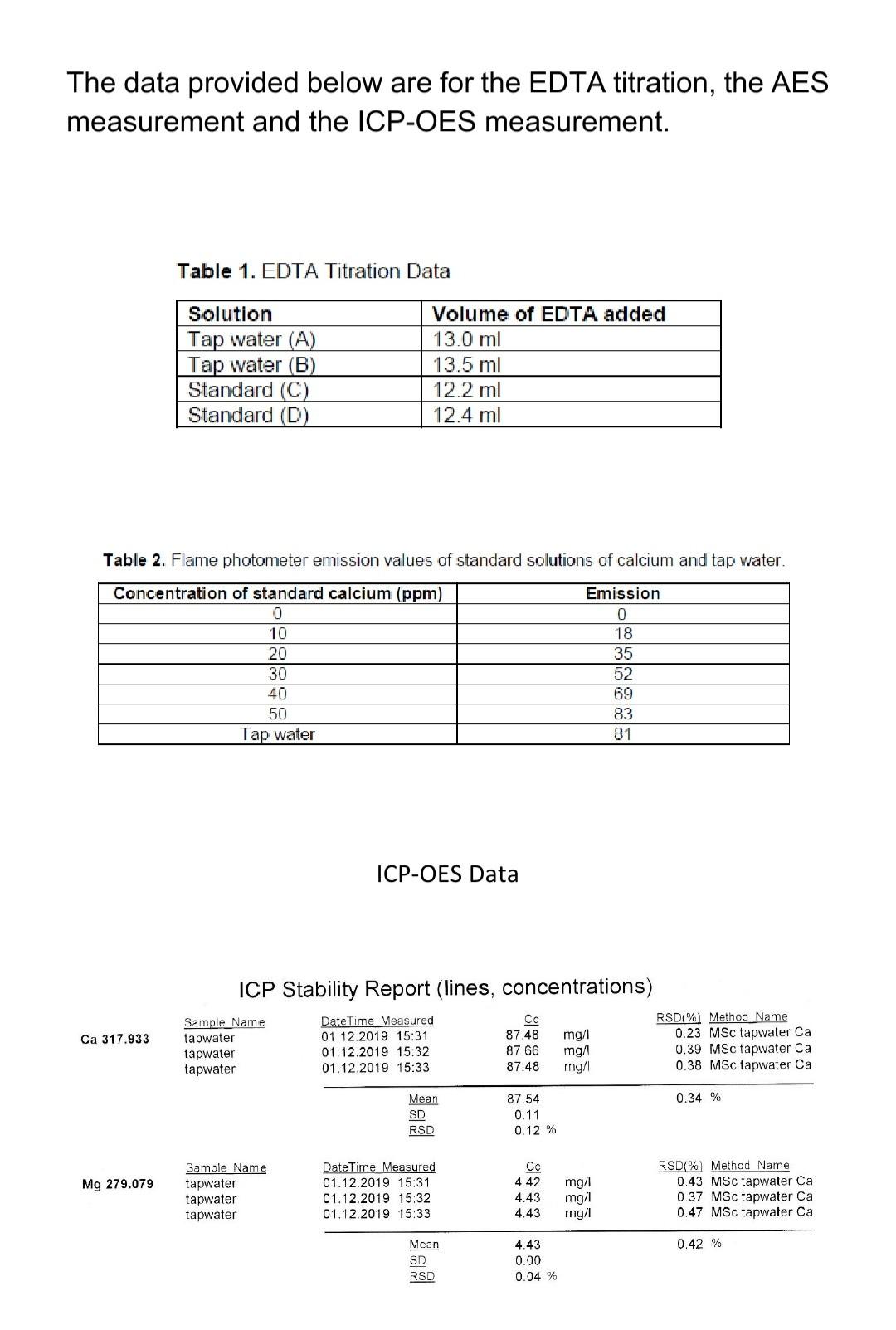
To Determine total hardness of Water sample in terms of Caco3 by EDTA Titration method using Eriochrome black T indicator – Chemical Practical – Go Practicals
Atomic Absorption Spectrophotometric and Ethylenediaminetetraacetate- Titration Methods for Calcium and Magnesium Determinations1

PDF) Indicator for the titrimetric determination of calcium and total calcium plus magnesium with ethylenediaminetetraacetate in water
Simultaneous determination of calcium, magnesium, and alkalinity by complexometric titration with potentiometric or photometric

SOLVED: 15-4 Pre-Lab Preparation Determining Ca? + and Mg? + by EDTA Titration: In this experiment, You will measure the concen- trations of Cal+ and Mg?- by two sets of titrations, using