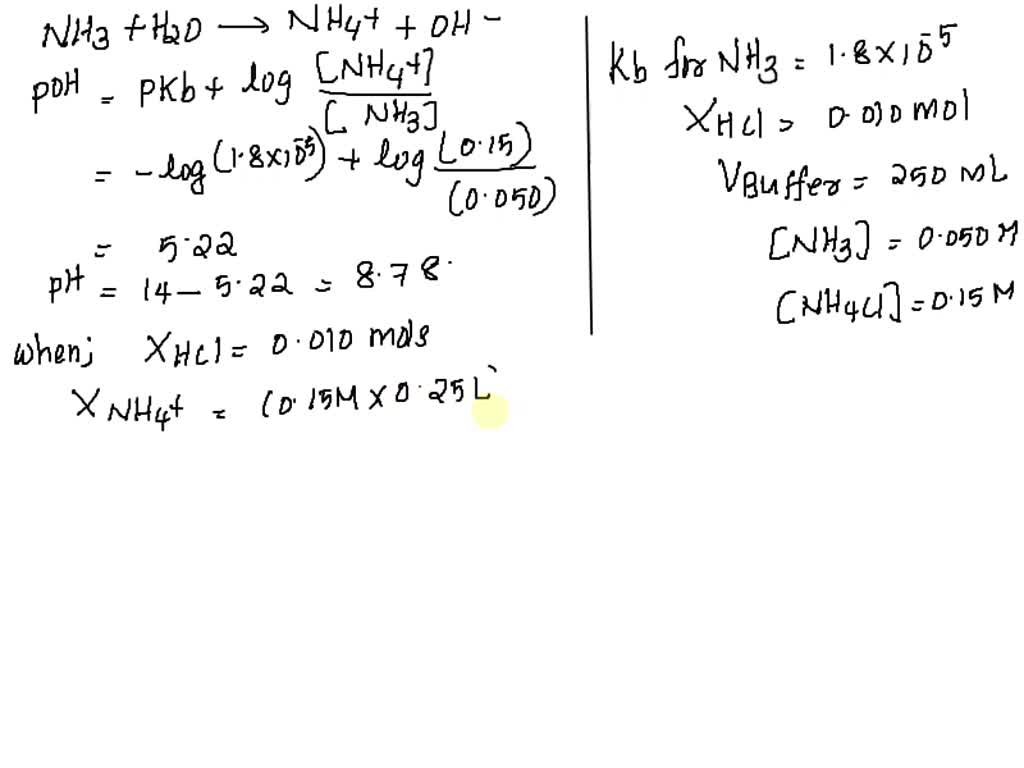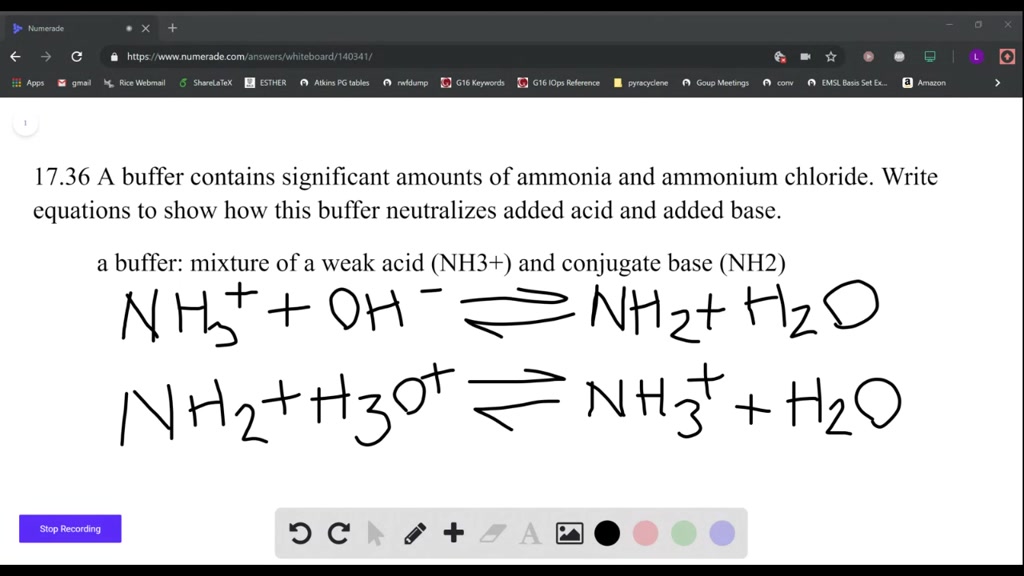
SOLVED: A buffer contains significant amounts of ammonia, nh3 , and ammonium chloride, nh4cl . part a write an equation showing how this buffer neutralizes added acid, hi.

SOLVED: Calculate the pH of a buffer that is 0.020 M in NH3 and 0.030 M in NH4Cl. What is the pH after adding 1.00 mL of 0.01 M NaOH to 0.10

OneClass: Calculate the pH of a .20M NH3/.20M NH4Cl buffer after the addition of 20.0mL of 0.10M HCl ...

a buffer solution contain NH3 and NH4CL total concentration of buffering agent is 0 6 molar if the pOH of - Chemistry - Equilibrium - 14195039 | Meritnation.com

A buffer solution made with NH3 and NH4Cl has a pH of 10.0 which procedure could be used to lower the pH ? 1.Adding HCl , 2.Adding NH3 , 3. Adding NH4Cl

Calculate the pH of 0.5 L of a 0.2 M NH4Cl - 0.2 M NH3 buffer before and after addition of (a) 0.05 mole of NaOH and (b) 0.05 mole of HCI.

pH calculation of a buffer solution made from a weak base and its conjugate acid (salt form) - YouTube
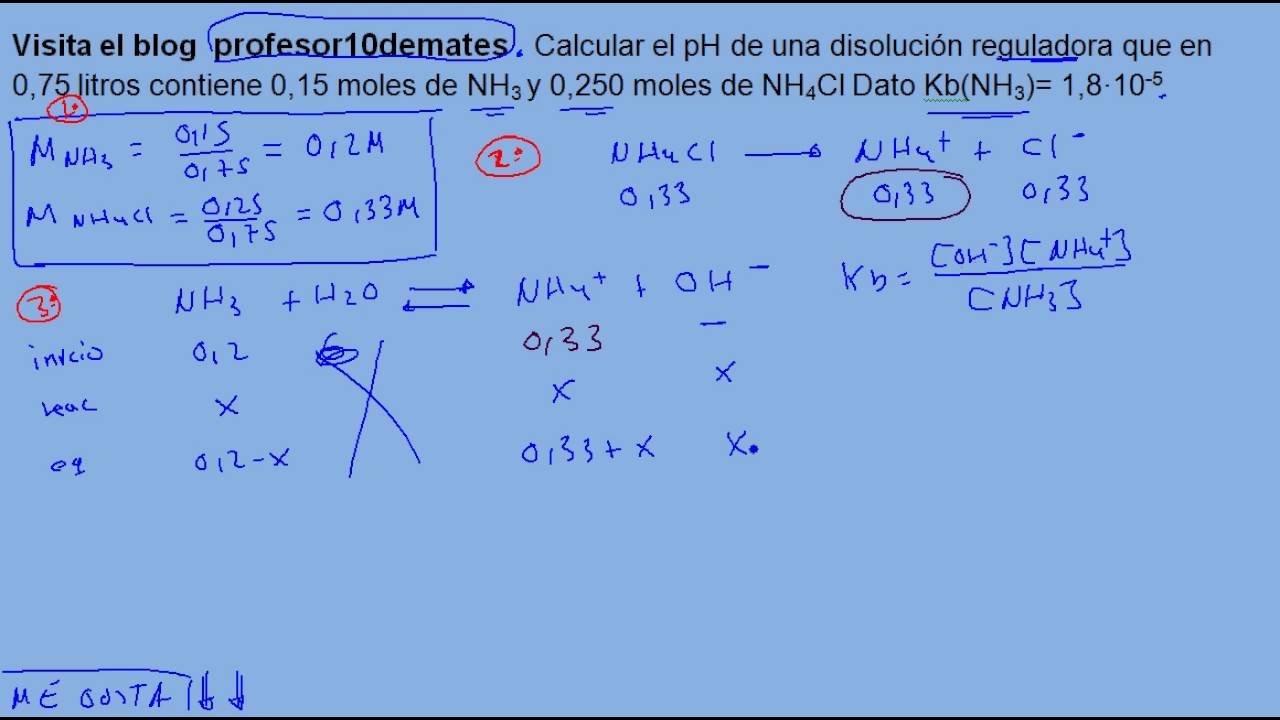
Calculate the amount of NH3 and NH4Cl required to prepare a buffer solution of pH = 9 when total concentration of buffering - Sarthaks eConnect | Largest Online Education Community

Calculate the amount of NH3 and NH4Cl required to prepare a buffer solution of pH 9.0 when total concentration of buffering reagents is 0.6 mol L^- . pKb for NH3 = 4.7, log 2 = 0.30 .